Its 6am, another orange-soaked spring morning in 2008, Nanjing, China.
I’m 3 feet away, gazing at an elderly gentleman who is upside down, supported only by the tip of his skull, arms crossed, legs delicately wrapped around a vertical bar. I learn that he is 84 years old, and that he often likes to hang upside down (for up to 3 hours); exercise parks, trees, rings…he tells me he often stands on one leg for 45 minutes at a time. He also informed me that for his 83rd birthday he spent the month cycling the 730 miles from Nanjing to Hong Kong, just to visit family. One of his elderly compatriots next to him is warming up into a full front splits position before she goes to see the grandkids. By now I’d come to understand that this behaviour is not anomalous amongst the geriatric population of China, but I’m still arrested by this man’s exploits and vitality.

Fast forward eleven years and I am now working as a junior doctor in a care of the elderly ward in the North of England. Here, witnessing a patient of the same age who is able to ‘independently mobilise without assistance’ to the toilet next to the nurse’s desk can be a rarity. Many of these elderly are cachectic, bed-bound, frail, or just ‘not coping’ (euphemistically known as ‘acopia’ in the profession).
Two 84 year old individuals, imagine twins, can begin ostensibly with the same phenotype and baseline, but eventually diverge to such a degree that one is house-bound and another is cycling to Hong Kong.
How does this happen?
Lets first speak briefly of ageing. Like death, ageing is the distant uncle that no-one wants to think about or comprehend. Like death, its also the uncle that will, absolutely, visit you when you are least prepared, have shit to do, and just want him to leave; except he never bloody leaves. Ever.
Despite successfully prolonging average human life expectancy (largely through sanitation, vaccinations, antibiotics), we have been unable to extend the limit of the human lifespan much past 115 years [1], which begs the question; what causes ageing?
Multiple mechanisms have been elaborated over recent decades [2]: free radical theory, which posits that a chronic accumulation of biological by-products (reactive oxygen species) wreaks damage to the body over time. An extension of this theory is that mitochondrial DNA, which codes for the power-houses of aerobic energy production inside the cell, gathers increasing rates of genetic mutations, leading to a snowballing leakage of reactive oxygen species, colliding with and damaging biological machinery across the body. ‘Inflam-ageing’ refers to the chaotic inflammatory landscape that can accompany the ageing body, caused by derangements such as increasing visceral fat and concurrent loss of lean muscle tissue. Similarly, telomeres, immunology, the neuroendocrine axis, gene regulation and other characters all have their own roles to play in the ongoing theatrical tragedy of entropy that leads to our demise.
Is there such a thing as ‘healthy’ ageing?
We live in a time where we are privileged enough to even ponder the concept of ‘healthy ageing’. Human history is brutally characterised by disease and war, and it has been difficult to tease out ‘healthy/natural ageing’ from the many pathologies and disease states that tend to accompany this; are these actually just an integral part of ‘healthy ageing’, or is there some independent pathology-free process? There have been a whole array of biomarkers proposed throughout the literature for tracking this [3]; grip strength, gait speed, chair rise, verbal fluency, blood pressure, lipids, bone mineral density, muscle mass, testosterone, IGF-1, IL-6…the list goes on. The point is, ageing is an all-encompassing, global phenomenon that affects virtually all bodily systems, and whilst we can aggregate individual metrics, sometimes a more unified and ‘spot-diagnosis’ approach can be helpful and intuitive.
People have talked about looking to lifelong athletes as the model for optimal, or ‘healthy’, ageing [4]. The average joe has likely already picked up some ailments along the lifespan, but masters athletes, who have continued to train, optimise recovery, receive nutritional and lifestyle support, and strive for ongoing personal best performances, are perhaps the best comparative model we have for what a ’natural’ ageing trajectory may look like. If we take a sport like powerlifting or gymnastics, this acts as an excellent proxy for global physical health and function. For instance, performing a world-record back squat as an ‘elderly’ adult tells us a lot about the overall biological state of such an individual.
At the time of this writing, Susan Elwyn, USA, is the 72 year old female who holds the deadlift world record of 130kg for the 52kg bodyweight class in the over-70’s category. Its not difficult to appreciate the likely dramatic difference in overall biological health that separates Elwyn from many of her similarly aged counterparts, who may struggle to stand up from a chair, whilst she can do so with 100kg (near double her bodyweight) on her back.

Dr Catherine Walter (71, left) of Oxford University Powerlifting Club alongside a more common form of geriatric squat (right)
Looking at the serial performances of these model individuals over time, one thing is certain: no matter how optimised an athlete’s life and training, there is an undeniable downward slope in performance for every individual and, by association, an unavoidable degradation of biology that occurs, independent of disease or pathology.

The interesting question is, how can you manipulate your inevitable downward slope?
Though this may sound bleak, entertain two potential scenarios: two people may die at exactly the same age of 80. One enjoys an independent life, fully mobile, able to pick up his grandkids, awake and energetic, until a lethal heart attack during a swing on the golf course (which describes my grandfather). The other same-aged individual may spend the preceding decades suffering falls, hyperglycaemia, cramping angina, shortness of breath on walking to the bathroom, chronic pain, and other miseries before a drawn out demise on a hospital ward.
These contrasting examples may sound extreme, but represent real-life dichotomies, the latter of which I currently see virtually every day. The concept that they illustrate is ‘compression of morbidity’; to squeeze chronic disease and ill-health into the smallest possible sliver at the end of one’s life, rather than suffer for a protracted period before one’s death. So how can you maximise the compression of your own morbidity?
Moving upstream
Being hit with the label of type 2 diabetes, ischaemic heart disease, hyperlipidaemia, osteoporosis, or many of the other non-communicable diseases may feel like a sudden shock, but the truth is that these maladies are in their infancy even decades before a clinical diagnosis. What may feel like ‘i’m alright’ now might overlook the current burgeoning insulin resistance, fatty liver, loss of beta cells, and metabolic chaos that will eventually, perhaps decades later, hit clinical threshold when the body is overwhelmed, when you hear the words ’type 2 diabetes’ in the clinic [5].
This is increasingly the health war that humans fight across the planet; its not smallpox, typhoid, gangrene, or other diseases of the ‘old-world’ that are likely to do you in. Its overwhelmingly the insidious, creeping, unfelt grip of chronic disease that stealthily begins its work in the background, until symptoms manifest and you have a formal diagnosis that requires medical intervention.
The current majority approach to the treatment of the preventable diseases is to rely on pharmeceutical therapy after onset of the disease…[4]
Four Geriatric Horsemen
Dr Jonathan Sullivan (MD), an Emergency physician and strength coach in Michigan, has coined the term ‘sick ageing phenotype’ [6] to describe the constellation of syndromes that most typically afflict us as we age: metabolic syndrome, sarcopenia and osteopenia, and polypharmacy. Although polypharmacy deserves real attention, for the now, I will exchange it for frailty, giving us ‘four horsemen of the ageing apocalypse’.

METABOLIC SYNDROME
Metabolic syndrome is typified by high blood pressure, dyslipidaemia, insulin resistance, and visceral adiposity. ‘Bla bla bla’ would be a fair response to this sentence, so let me try and explain what the crack is here. I was surprised to learn that fat tissue is in fact an endocrine organ, i.e. it actually produces hormones that affect the entire body, such as leptin, adiponectin, and resistin. Furthermore, visceral adiposity, the fat that surrounds your organs, is highly pro-inflammatory, releasing factors like IL-6, TNF-a and other cytokines that promote inflammation [7].
The problem with ongoing levels of inflammation is that tissue damage accrues: blood vessels expand, they become more permeable, immune cells (neutrophils, lymphocytes, macrophages) get through and infiltrate tissues, they release granules, proteases and other nasties that lead to tissue breakdown, which is then ‘repaired’ via scar tissue formation [8]. This is how atherosclerosis (clogged arteries) forms, setting the stage for a disabling stroke or lethal heart attack. Chronic inflammation is also associated with cancers, joint conditions, fatty liver, depression, and too many more to list. So its no bloody joke when fat tissue levels remain high.
Insulin resistance is a major component of metabolic syndrome. The government-recommended food pyramid doesn’t help the situation, and most of us are eating far more carbohydrates than we could possibly need. I’ll try to summise this issue as best I can using a sweet, sweet brownie. Brownies have a lot of carbohydrates. Carbohydrates (except fibre) are all eventually broken down to glucose (fructose undergoes different metabolism), the simplest carb, for the body to use. When you take a bite of that (delicious) brownie, insulin is immediately released in anticipation. Meanwhile, enzymes in the mouth and intestine are breaking down that brownie into glucose, absorbing it through the gut, and eventually through the liver into the blood stream. The body only likes a very specific amount of glucose in the bloodstream for various reasons.
So what does insulin do? Primarily, two major things:
- shuttle glucose the hell out of the blood and into cells for the body to use and
- store energy by converting glucose into adipose (fat) tissue for use later (yeah, right).
Ok, let’s say I keep pounding those brownies (yesss). More carbs coming in, more insulin needed to get glucose out of my blood and into the cells, as well as to store it for later as fat. More fat is accruing in muscle tissue, around organs, around the waist, and even within the liver (hence fatty liver disease development). At the same time, insulin is being churned out to try and sort all this glucose out. Soon, cells everywhere becomes less sensitive to the insulin by down regulating GLUT-4 receptors (think eventually tuning out the kid who is screaming in the background all the time), and so even more insulin is produced.
This goes on for years. Eventually, the pancreas just cannot keep this up, and the system is overwhelmed, the balance is lost, and glucose levels rise. One blood test later and you now have type 2 diabetes. Not to mention all the inflammatory chaos that accompanied getting here.

High blood pressure and messed up blood lipid (fat) levels have also been along for the ride, damaging the lining of arteries, causing them to become more stiff and less compliant, and threatening to damage end organs as well as heart and brain.
So that’s a bit about metabolic syndrome. Next up, and arguably further down the line, are the ugly siblings of sarcopenia and osteopenia.
SARCOPENIA AND OSTEOPENIA
Sarcopenia is the loss of muscle tissue, strength, and performance. In fact, in the same way that we have heart, liver and kidney failure, working groups worldwide have agreed that sarcopenia is a disease of ‘muscle failure’ [9]. Increasing age and decreasing muscular exertion results in predominant loss of type II fibres (the highest force producing fibres), denervation of motor units, atrophy and loss of muscle cells, and a subsequent decrease in physical function.
Aside from this, the problem of inflamation once again rears its ugly head; muscle tissue, through the process of contraction, produces anti-inflammatory cytokines (myokines – from the muscle) which in turn suppress pro-inflammatory factors throughout the body. Without this counter-regulatory force that the muscles provide, the system tilts and then slides rapidly toward a pro-inflammatory state. Furthermore, low levels of muscle tissue mean a small pool of protein and amino acids for other organs to draw from in their own ongoing maintenance and repair. In addition, muscle tissue, which usually makes up around 40% of total bodyweight in a non-sarcopenia individual, is also the single biggest disposal system for glucose in the body. And so the viscous cycle of reduced muscle —> reduced glucose disposal —> hyperglycaemia and insulin resistance, continues to accelerate.
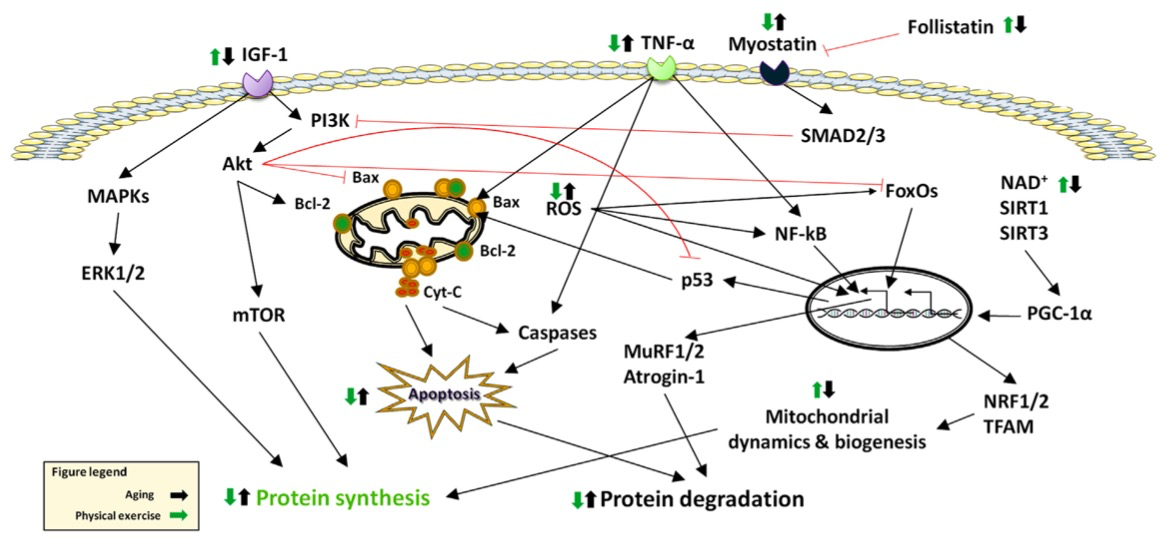
Osteopenia is the reduction of bone mineral density, -1 to -2.5 standard deviations from the population mean. Bone mineral density reduction is a natural phenomenon of age, particularly sped up for women after the menopause when oestrogen levels drastically decline. However, drifting too far into this territory, primarily from a lack of vitamin D and loaded activity (both of which can be easily prescribed), can predispose us to unexpected fractures.
I have met numerous patients who, whilst undertaking something as innocuous as walking to their kitchen, suddenly experienced a sharp pain in their mid back when their vertebral body spontaneously wedge-fractured, simply from poor bone mineral density. These carry the grim title of ‘osteoporotic fragility fractures’. Hip fractures are the same deal, and occupy 1% of the entire NHS budget, with 6.9% of people dying within 30 days of such an event [10]. Unfortunately, the popularly recommended activities of non-weight bearing exercise such as swimming and cycling do nothing attenuate or prevent this.
FRAILTY
Whilst sarcopenia is a defined disease (muscle failure), frailty describes a syndrome that is more all-encompassing and reflective of actual physical function. Although there are a number of different criteria and definitions, the unifying concept underpinning frailty is that of having poor reserve; physiological and cognitive.
Think of reserve as the capacity to buffer stressors, or an alternative term may be resilience. A 21 year old will generally have ample resilience to recover quickly and back to their previous self from most insults that may land them in hospital. Conversely, the reserve, or resources, of a frail older adult may be virtually exhausted and unrenewable after one bout of pneumonia. Even if they do manage to recover from the illness, their reserve and function may have taken a substantial stepwise hit, lowering their threshold to subsequent threats.

From an ‘eyeballing’ level, frailty carries features of weakness, slowness, exhaustion, weight loss, poor cognition, and low levels of physical activity . From a ‘microscope’ level, there are a disturbing number of crossovers between frailty with our other geriatric horsemen mentioned above. Large cohort older studies have confirmed higher levels of inflammatory markers such as TNF-a, IL-6 and CRP in frailer groups, which as we remember, lead to systemic tissue damage, as well as likely contributing to sarcopenia, anaemia, and legion of disease states [11].
Frailty also predicts increased falls, hospitalisation, longer hospital stays, dependance and mortality. As for its treatment, testosterone and oestrogen replacement therapy, IGF-1, and vitamin D/calcium supplementation may all have their place, but as per the thrust of this article, authors have noted:
To date, exercise is the interventional modality that has most consistently shown benefit in treating frailty and its key components [11]
The upshot of all this misery listed above is that, incredibly, it is largely preventable and treatable. The antidote isn’t anything you can buy, its not a procedure, its not offered by big pharma; it actually lies dormant in the ancient wisdom of your own musculature and biological machinery. You have an endogenous pharmacy at your disposal, without side-effects, and exquisitely titrated to your own unique phenotype.
And that carries us along to part II of this topic, where we will discuss how to access and accrue the potential lifelong vitality and therapeutic benefits available to each of us.
[DISPLAY_ULTIMATE_SOCIAL_ICONS]

References:
- Vijg J, Le Bourg E. Aging and the Inevitable Limit to Human Life Span. Gerontology. 2017;63(5):432–4.
- Tosato M, Zamboni V, Ferrini A, Cesari M. The aging process and potential interventions to extend life expectancy. Clin Interv Aging [Internet]. 2007;2(3):401–12. Available from: http://www.ncbi.nlm.nih.gov/pubmed/18044191 http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=PMC2685272
- Mathers J, Deary I, Kuh D, Lord J, Khaw K-T, Lara J, et al. Guidelines for biomarkers of healthy ageing. Guidel biomarkers Heal ageing [Internet]. 2015;93. Available from: http://www.mrc.ac.uk/documents/pdf/biomarkers-of-healthy-ageing/
- Lazarus NR, Harridge SDR. The inherent human aging process and the facilitating role of exercise. Frontiers in Physiology. 2018.
- Sagesaka H, Sato Y, Someya Y, Tamura Y, Shimodaira M, Miyakoshi T, Hirabayashi K, Koike H, Yamashita K, Watada H, Aizawa T. Type 2 Diabetes: When Does It Start? J Endocr Soc. 2018 Apr 18;2(5):476-484. doi: 10.1210/js.2018-00071. PubMed PMID: 29732459; PubMed Central PMCID: PMC5932476.
- Sullivan JM, Baker A. The Barbell Prescription; Strength Training for Life After 40, The Aasgaard Company, Dec 2016.
- Galancho-Reina I, Sanchez-Oliver JS, Gonzalez-Matarin PJ, Butragueno J, Bandera-Merchan B, Suarez-Carmona W, Isidro-donate F, Tinahones FJ, Macias-Gonzalez M. The Role of Muscle Tissue and Resistance Training in Cardiometabolic Health. Int J Sport Science & Medicine. Jan 2019. ISSN: 2640-0936
- Pahwa R, Jialal I. Chronic Inflammation. [Updated 2018 Oct 27]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2019 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK493173/
- Cruz-Jentoft AJ, Bahat G, Bauer J, Boirie Y, Bruyère O, Cederholm T, et al. Sarcopenia: Revised European consensus on definition and diagnosis. Age and Ageing. 2019. p. 16–31.
- Royal College of Physicians. National Hip Fracture Database (NHFD) Annual Report. Royal College of Physicians. 2017.
- Chen X, Mao G, Leng SX. Frailty syndrome: An overview. Clinical Interventions in Aging. 2014. p. 433–41.
- Pratesi A, Tarantini F, Di Bari M. Skeletal muscle: An endocrine organ. Clinical Cases in Mineral and Bone Metabolism. 2013. p. 11–4.
- Welch C, K. Hassan-Smith Z, A. Greig C, M. Lord J, A. Jackson T. Acute Sarcopenia Secondary to Hospitalisation – An Emerging Condition Affecting Older Adults. Aging Dis [Internet]. 2018;9(1):151. Available from: http://www.aginganddisease.org/EN/10.14336/AD.2017.0315
- Ziaaldini MM, Marzetti E, Picca A, Murlasits Z. Biochemical Pathways of Sarcopenia and Their Modulation by Physical Exercise: A Narrative Review. Front Med. 2017;4.
- 1. Siparsky PN, Kirkendall DT, Garrett WE. Muscle Changes in Aging: Understanding Sarcopenia. Sports Health. 2014. p. 36–40.


Leave a Reply
You must be logged in to post a comment.